How-To?
Cheese Production


For most cheeses produced worldwide, cow’s milk is used, however milk of other animals, especially goat and sheep is also widely used. The quality of milk used in (semi-) industrial cheese making is rigidly controlled in Europe. The majority of cheeses are made from heat-treated or pasteurised milk (either whole, low-fat or non-fat). If non-pasteurised milk is used, the cheese must be ripened for at least 60 days at a temperature of not less than 4°C to ensure safety against pathogenic organisms. Pasteurisation requirements for milk used to make specific cheese varieties are regulated differently in each country.
Cheese making involves a number of main stages that are common to most types of cheese.
The cheese milk is pre-treated, possibly preripened after addition of a bacteria culture appropriate to the type of cheese, and mixed with rennet.
The enzyme activity of the rennet causes the milk to coagulate to a solid gel known as coagulum or curd. This is cut with special cutting tools into small cubes of the desired size – in the first place to facilitate expulsion of whey. During the rest of the curd making process the bacteria grow and form lactic acid, and the curd grains are subjected to mechanical treatment with stirring tools, while at the same time the curd is heated according to a preset programme.
The combined effect of these three actions – growth of bacteria, mechanical treatment and heat treatment – results in syneresis, i.e. separation of whey from the curd grains. The finished curd is placed in cheese moulds of metal, wood or plastic, which determine the shape of the finished cheese.
The cheese is pressed, either by its own weight or more commonly by applying pressure to the moulds. Treatment during curd making and pressing determines the characteristics of the cheese. The actual flavour of the cheese is determined during the ripening of the cheese.
Different steps in cheese making are discussed below.
Pasteurisation
Before the actual cheese making begins, the milk usually undergoes pre-treatment designed to create optimum conditions for production.
Milk intended for types of cheese which require more than one month for ripening need not necessarily be pasteurised, but usually is. Milk intended for unripened cheese (fresh cheese) must be pasteurised.
Milk intended for original Emmenthal, Parmesan and Grana, some extra hard types of cheese, must not be heated to more than 40°C, to avoid affecting flavour, aroma and whey expulsion. Milk intended for these types of cheese normally comes from selected dairy farms with frequent veterinary inspection of the herds.
Although cheese made from unpasteurised milk is considered to have a better flavour and aroma, most producers (except makers of the extra hard types) pasteurise the milk because its quality is seldom so dependable that they are willing to take the risk of not pasteurising it.
Pasteurisation must be sufficient to kill bacteria capable of affecting the quality of the cheese, e.g. coliforms, which can cause early “blowing” and a disagreeable taste. Regular pasteurisation at 72 – 73°C for 15 – 20 seconds is most commonly applied.
However, spore-forming microorganisms in the spore state survive pasteurisation and can cause serious problems during the ripening process. One example is Clostridium tyrobutyricum, which forms butyric acid and large volumes of hydrogen gas by fermenting lactic acid. This gas destroys the texture of the cheese completely (‘blowing’), not to mention the fact that butyric acid is unsavoury.
More intense heat treatment would reduce that particular risk, but would also seriously impair the general cheese making properties of the milk. Other means of reducing thermotolerant bacteria are therefore used.
Traditionally, certain chemicals have been added to cheese milk prior to production to prevent “blowing” and development of the unpleasant flavour caused by heat-resistant spore-forming bacteria (principally Clostridium tyrobutyricum). The most commonly used chemical is sodium nitrate (NaNO3), but at production of Emmenthal cheese, hydrogen peroxide (H2O2) is also used. However, as the use of chemicals has been widely criticised, mechanical means of reducing the number of unwanted microorganisms have been adopted, particularly in countries where the use of chemical inhibitors is banned.
Starter cultures
The starter culture is a very important factor in cheese making; it performs several duties.
Two principal types of culture are used in cheese making:
– mesophilic cultures with a temperature optimum between 20 and 40°C
and
– thermophilic cultures which develop at up to 45°C.
The most frequently used cultures are mixed strain cultures, in which two or more strains of both mesophilic and thermophilic bacteria exist in symbiosis, i.e. to their mutual benefit. These cultures not only produce lactic acid but also aroma components and CO2. Carbon dioxide is essential to creating the cavities in round-eyed and granular types of cheese. Examples are Gouda, Manchego and Tilsiter from mesophilic cultures and Emmenthal and Gruyère from thermophilic cultures.
Single-strain cultures are mainly used where the object is to develop acid and contribute to protein degradation, e.g. in Cheddar and related types of cheese.
Three characteristics of starter cultures are of primary importance in cheese making, viz.
- ability to produce lactic acid in the curd
- ability to break down the protein and, when applicable,
- ability to produce carbon dioxide (CO2).
When milk coagulates, bacterial cells are concentrated in the coagulum and thus in the cheese.
Development of acid lowers the pH, which is important in assisting syneresis (contraction of the coagulum accompanied by elimination of whey).
Furthermore, salts of calcium and phosphorus are released, which influence the consistency of the cheese and help to increase the firmness of the curd.
Another important function performed by the acid-producing bacteria is to suppress surviving bacteria after pasteurisation or recontamination bacteria. The latter need lactose, which is converted by the starter bacteria and is thus no longer available for growth. The lactic acid formed is also a anti-microbial factor.
Production of lactic acid stops when all the lactose in the cheese (except in soft cheeses) has been fermented. Lactic acid fermentation is normally a relatively fast process. In some types of cheese, such as Cheddar, it must be completed before the cheese is pressed, and in other types within a week.
If the starter also contains CO2-forming bacteria, acidification of the curd is accompanied by production of carbon dioxide through the action of citric acid fermenting bacteria. Mixed strain cultures with the ability to develop CO2 are essential for production of cheese with a texture with round holes/eyes or irregularly shaped eyes. The evolved gas is initially dissolved in the moisture phase of the cheese; when the solution becomes saturated, the gas is released and creates the eyes.
The ripening process in hard and certain semi-hard cheeses is a combined proteolytic effect where the original enzymes of the milk and those of the bacteria in the culture, together with rennet enzyme, cause decomposition of the protein.
Other additions before making the curd
Calcium chloride (CaCl2)
If the milk is of poor quality for cheese making, the coagulum will be soft. This results in heavy losses of fines (casein) and fat as well as poor syneresis during cheese making.
5 – 20 grams of calcium chloride per 100 kg of milk is normally enough to achieve a constant coagulation time and result in sufficient firmness of the coagulum. Excessive addition of calcium chloride may make the coagulum so hard that it is difficult to cut.
For production of low-fat cheese, and if legally permitted, disodium phosphate (Na2PO4), usually 10 – 20 g/kg, can sometimes be added to the milk before the calcium chloride is added. This increases the elasticity of the coagulum due to formation of colloidal calcium phosphate (Ca3(PO4)2), which will have almost the same effect as the milk fat globules entrapped in the curd.
Carbon dioxide (CO2)
Addition of CO2is one method of improving the quality of cheese milk. Carbon dioxide occurs naturally in milk, but most of it is lost in the course of processing. Adding carbon dioxide by artificial means lowers the pH of the milk: the original pH is normally reduced by 0.1 to 0.3 units. This will then result in shorter coagulation time. The effect can be utilised to obtain the same coagulation time with a smaller amount of rennet.
Saltpetre (NaNO3 or KNO3)
Fermentation problems may be experienced if the cheese milk contains butyric-acid bacteria (Clostridia) and/or coliform bacteria.
Saltpetre (sodium or potassium nitrate) can be used to counteract these bacteria, but the dosage must be accurately determined with reference to the composition of the milk, the process for the type of cheese, etc., as too much saltpetre will also inhibit growth of the starter. Overdosage of saltpetre may affect the ripening of the cheese or even stop the ripening process.
Saltpetre in high doses may discolour the cheese, causing reddish streaks and an impure taste. The maximum permitted dosage is about 30 grams of saltpetre per 100 kg of milk.
In the past decade usage of saltpetre has been questioned from a medical point of view, and in some countries it is also forbidden.
Colouring agents
The colour of cheese is to a great extent determined by the colour of the milk fat, and undergoes seasonal variations. Colours such as carotene and orleana, a natural anatto dye, are used to correct these seasonal variations in countries where colouring is permitted.
Green chlorophyll (contrast dye) is also used, for example for blueveined cheese, to obtain a “pale” colour as a contrast to the blue mould.
Rennet
Except for types of fresh cheese such as cottage cheese and quarg, in which the milk is clotted mainly by lactic acid, all cheese manufacture depends upon formation of curd by the action of rennet or similar enzymes.
Coagulation of casein is the fundamental process in cheese making. It is generally done with rennet, but other proteolytic enzymes can also be used, as well as acidification of the casein to the iso-electric point (pH 4.6 – 4.7).
The active principle in rennet is an enzyme called chymosine, and coagulation takes place shortly after the rennet is added to the milk. There are several theories about the mechanism of the process, and even today it is not fully understood. However, it is evident that the process operates in several stages; it is customary to distinguish these as follows:
- Transformation of casein to paracasein under the influence of rennet
- Precipitation of paracasein in the presence of calcium ions.
The whole process is governed by the temperature, acidity, and calcium content of the milk as well as other factors. The optimum temperature for rennet is in the region of 40°C, but lower temperatures are normally used in the practice, basically to avoid excessive hardness of the coagulum.
Rennet is extracted from the stomachs of young calves and marketed in form of a solution with a strength of 1:10 000 to 1:15 000, which means that one part of rennet can coagulate 10 000 – 15 000 parts of milk in 40 minutes at 35°C. Bovine and porcine rennet are also used, often in combination with calf rennet (50:50, 30:70, etc.). Rennet in powder form is normally 10 times as strong as liquid rennet.
Substitutes for animal rennet
About 50 years ago, investigations were started to find substitutes for animal rennet. This was done primarily in India and Israel on account of vegetarians’ refusal to accept cheese made with animal rennet. In the Muslim world, the use of porcine rennet is out of the question, which is a further important reason to find adequate substitutes. Interest in substitute products has grown more widespread in recent years due to a shortage of animal rennet of good quality.
There are two main types of substitute coagulants:
- Coagulating enzymes from plants,
- Coagulating enzymes from micro-organisms.
Investigations have shown that coagulation ability is generally good with preparations made from plant enzymes. A disadvantage is that the cheese very often develops a bitter taste during storage.
Various types of bacteria and moulds have been investigated, and the coagulation enzymes produced are known under various trade names. DNA technology has been utilised in recent times, and a DNA rennet with characteristics identical to those of calf rennet is now being thoroughly tested with a view to securing approval.
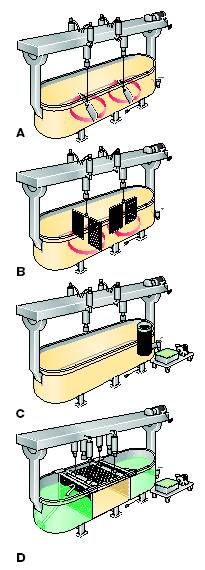 |
Example of a conventional cheese vat at different stages:
A : during stirring B : during cutting C : during whey drainage D : during pressing Source : |
Cutting the coagulum
The renneting or coagulation time is typically about 30 minutes. Before the coagulum is cut, a simple test is normally carried out to establish its whey eliminating quality. Typically, a knife is stuck into the clotted milk surface and then drawn slowly upwards until proper breaking occurs. The curd may be considered ready for cutting as soon as a glass-like splitting flaw can be observed. Cutting gently breaks the curd up into grains with a size of 3 – 15 mm depending on the type of cheese. The finer the cut, the lower the moisture content in the resulting cheese.
Pre-stirring
Immediately after cutting, the curd grains are very sensitive to mechanical treatment, for which reason the stirring has to be gentle. It must however be fast enough to keep the grains suspended in the whey. Sedimentation of curd in the bottom of the vat causes formation of lumps. This puts strain on the stirring mechanism, which must be very strong. The curd of low fat cheese has a strong tendency to sink to the bottom of the vat, which means that the stirring must be more intense than for curd of high fat content. Lumps may influence the texture of the cheese as well as causing loss of casein in whey.
Pre-drainage of whey
For some types of cheese, such as Gouda and Edam, it is desirable to rid the grains of relatively large quantities of whey so that heat can be supplied by direct addition of hot water to the mixture of curd and whey, which also lowers the lactose content. Some producers also drain off whey to reduce the energy consumption needed for indirect heating of the curd. For each individual type of cheese it is important that the same amount of whey – normally 35%, sometimes as much as 50% of the batch volume – is drained off every time.
Heating/cooking/scalding
Heat treatment is required during cheese making to regulate the size and acidification of the curd. The growth of acid-producing bacteria is limited by heat, which is thus used to regulate production of lactic acid. Apart from the bacteriological effect, the heat also promotes contraction of the curd accompanied by expulsion of whey (syneresis).
Depending on the type of cheese, heating can be done in the following ways:
- By steam in the vat/tank jacket only.
- By steam in the jacket in combination with addition of hot water to the curd/whey mixture.
- By hot water addition to the curd/whey mixture only.
The time and temperature programme for heating is determined by the method of heating and the type of cheese. Heating to temperatures above 40°C, sometimes also called cooking, normally takes place in two stages. At 37 – 38°C the activity of the mesophilic lactic acid bacteria is retarded, and heating is interrupted to check the acidity, after which heating continues to the desired final temperature. Above 44°C the mesophilic bacteria are totally deactivated, and they are killed if held at 52°C between 10 and 20 minutes.
Heating beyond 44°C is typically called scalding. Some types of cheese, such as Emmenthal, Gruyère, Parmesan and Grana, are scalded at temperatures as high as 50 – 56°C. Only the most heat-resistant lactic-acid-producing bacteria survive this treatment. One that does so is Propionibacterium freudenreichii ssp. shermanii, which is very important to the formation of the character of Emmenthal cheese.
Final stirring
The sensitivity of the curd grains decreases as heating and stirring proceed. More whey is exuded from the grains during the final stirring period, primarily due to the continuous development of lactic acid but also by the mechanical effect of stirring.
The duration of final stirring depends on the desired acidity and moisture content in the cheese.
Final removal of whey and principles of curd handling
As soon as the required acidity and firmness of the curd have been attained – and checked by the producer – the residual whey is removed from the curd in various ways depending on the type of cheese.
Cheese with granular texture
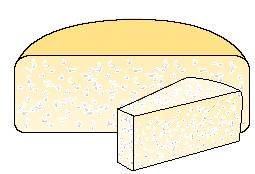
One-way is to withdraw whey direct from the cheese vat; this is used mainly with manually operated open cheese vats. After whey drainage the curd is scooped into moulds. The resulting cheese acquires a texture with irregular holes or eyes , also called agranular texture, see image above. The holes are primarily formed by the carbon dioxide gas typically evolved by so-called LD starter cultures (Lactococcus lactis, Leuconostoc cremoris and Lactococcus diacetylactis).
If curd grains are exposed to air before being collected and pressed, they do not fuse completely; a large number of tiny air pockets remain in the interior of the cheese. The carbon dioxide formed and released during the ripening period fills and gradually enlarges these pockets. The holes formed in this way are irregular in shape.
Whey can also be drained by pumping the curd/whey mixture across a vibrating or rotating strainer, where the grains are separated from the whey and discharged direct into moulds. The resulting cheese has a granular texture.
Round-eyed cheese

Gas-producing bacteria, similar as mentioned above are also used in production of round-eyed cheese, but the procedure is somewhat different.
According to older methods, e.g. for production of Emmenthal cheese, the curd was collected in cheese cloths while still in the whey and then transferred to a large mould on a combined drainage and pressing table. This avoided exposure of the curd to air prior to collection and pressing, which is an important factor in obtaining the correct texture in that type of cheese.
Studies of the formation of round holes/eyes have shown that when curd grains are collected below the surface of the whey, the curd contains microscopic cavities. Starter bacteria accumulate in these tiny whey-filled cavities. The gas formed when they start growing, initially dissolves in the liquid, but as bacteria growth continues, local supersaturation occurs which results in the formation of small holes. Later, after gas production has stopped due to lack of substrate, diffusion becomes the most important process. This enlarges some of the holes that are already relatively large, while the smallest holes disappear. Enlargement of bigger holes at the expense of the smaller ones is a consequence of the laws of surface tension, which state that it takes less gas pressure to enlarge a large hole than a small one.
Closed texture cheese
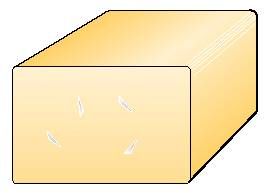
Closed texture types of cheese, of which Cheddar is a typical example, are normally made with starter cultures containing bacteria that do not evolve gas – typically single-strain lactic-acid-producing bacteria like Lactococcus cremonis and Lactococcus lactis .
The specific processing technique may however result in formation of cavities called mechanical holes. While the holes in granular and round-eyed cheeses have a characteristically shiny appearance, mechanical holes have rough inner surfaces.
When the acidity of the whey has reached about 0.2 – 0.22% lactic acid (about 2 hours after renneting), the whey is drained off and the curd is subjected to a special form of treatment called cheddaring. After all whey has been discharged, the curd is left for continued acidification and matting. During this period, typically 2 – 2.5 hours, the curd is formed into blocks which are turned upside down and stacked.
Final treatment of curd
As previously mentioned, the curd can be treated in various ways after all the free whey has been removed. It can be:
- transferred direct to moulds (granular cheeses),
- pre-pressed into a block and cut into pieces of suitable size for placing in moulds (round-eyed cheeses), or
- sent to cheddaring, the last phase of which includes milling into chips which can be dry-salted and either hooped or, if intended for Pasta Filata types of cheese, transferred unsalted to a cooking-stretching machine.
Pressing
After having been moulded or hooped the curd is subjected to final pressing, the purpose of which is fourfold:
- to assist final whey expulsion,
- to provide texture,
- to shape the cheese,
- to provide a rind on cheeses with long ripening periods.
The rate of pressing and pressure applied are adapted to each particular type of cheese. Pressing should be gradual at first, because initial high pressure compresses the surface layer and can lock moisture into pockets in the body of the cheese.
Salting
In cheese, as in a great many foods, salt normally functions as a condiment. But salt has other important effects, such as retarding starter activity and bacterial processes associated with cheese ripening. Application of salt to the curd causes more moisture to be expelled, both through an osmotic effect and a salting effect on the proteins. The osmotic pressure can be likened to the creation of suction on the surface of the curd, causing moisture to be drawn out.
With few exceptions, the salt content of cheese is 0.5 – 2%. Blue cheese and white pickled cheese variants (Feta, Domiati, etc.), however, normally have a salt content of 3 – 7%.
The exchange of calcium for sodium in paracaseinate that results from salting also has a favourable influence on the consistency of the cheese, which becomes smoother. In general, the curd is exposed to salt at a pH of 5.3 – 5.6 i.e. approx. 5 – 6 hours after the addition of a vital starter, provided the milk does not contain bacteria-inhibiting substances.
Dry salting
Dry salting can be done either manually or mechanically. Salt is applied manually from a bucket or similar container containing an adequate (weighed) quantity that is spread as evenly as possible over the curd after all whey has been discharged. For complete distribution, the curd may be stirred for 5 – 10 minutes.
There are various ways to distribute salt over the curd mechanically. One is the same as is used for dosage of salt on cheddar chips during the final stage of passage through a continuous cheddaring machine.
Brine salting
Brine salting systems of various designs are available, from fairly simple ones to technically very advanced ones. Still, the most commonly used system is to place the cheese in a container with brine. The containers should be placed in a cool room at about 12 – 14°C.
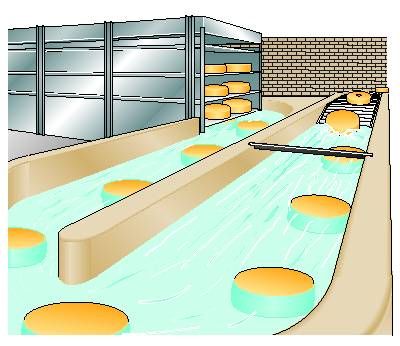
Industrial brine salting system. Source : Dairy Processing Handbook, Tetrapak Sweden
Salt content in different types of cheese
% salt
- Cottage cheese 0.25 – 1.0
- Emmenthal 0.4 – 1.2
- Gouda 1.5 – 2.2
- Cheddar 1.75 – 1.95
- Limburger 2.5 – 3.5
- Feta 3.5 – 7.0
- Gorgonzola 3.5 – 5.5
- Other blue cheeses 3.5 – 7.0
Ripening and storage of cheese
Ripening (curing)
After curdling all cheese, apart from fresh cheese, goes through a whole series of processes of a microbiological, biochemical and physical nature.
These changes affect both the lactose, the protein and the fat and constitute a ripening cycle which varies widely between hard, medium-soft and soft cheeses. Considerable differences occur even within these groups.
Lactose decomposition
The techniques which have been devised for making different kinds of cheese are always directed towards controlling and regulating the growth and activity of lactic acid bacteria. In this way it is possible to influence simultaneously both the degree and the speed of fermentation of lactose. It has been stated previously that in the process for making Cheddar, the lactose is already fermented before the curd is hooped. As far as the other kinds of cheese are concerned, lactose fermentation ought to be controlled in such a way that most of the decomposition takes place during the pressing of the cheese and, at latest, during the first week or possibly the first two weeks of storage.
The lactic acid which is produced is neutralised to a great extent in the cheese by the buffering components of milk, most of which have been included in the coagulum. Lactic acid is thus present in the form of lactates in the completed cheese. At a later stage, the lactates provide a suitable substrate for the propionic acid bacteria which are an important part of the microbiological flora of Emmenthal, Gruyère and similar types of cheese.
Besides propionic acid and acetic acid, considerable amounts of carbon dioxide are formed, which are the direct cause of the formation of the large round eyes in the above-mentioned types of cheese.
The lactates can also be broken down by butyric acid bacteria, if the conditions are otherwise favourable for this fermentation, in which case hydrogen is released in addition to certain volatile fatty acids and carbon dioxide. This faulty fermentation arises at a late stage, and the hydrogen can actually cause the cheese to burst.
Fermentation of lactose is caused by the lactase enzyme present in lactic acid bacteria.
Protein decomposition
The ripening of cheese, especially hard cheese, is characterised first and foremost by the decomposition of protein. The degree of protein decomposition affects the quality of the cheese to a very considerable extent, most of all its consistency and taste. The decomposition of protein is brought about by the enzyme systems of
- rennet
- micro-organisms
- plasmin, an protein degrading enzyme
The only effect of rennet is to break down the paracasein molecule into polypeptides. This first attack by the rennet, however, makes possible a considerably quicker decomposition of the casein through the action of bacterial enzymes than would be the case if these enzymes had to attack the casein molecule directly. In cheese with high cooking temperatures, scalded cheeses like Emmenthal and Parmesan, plasmin activity plays a role in this first attack.
In medium-soft cheeses like Tilsiter and Limburger, two ripening processes proceed parallel to each other, viz. the normal ripening process of hard rennet cheese and the ripening process in the smear which is formed on the surface. In the latter process, protein decomposition proceeds further until finally ammonia is produced as a result of the strong proteolytic action of the smear bacteria.
Storage
The purpose of storage is to create the external conditions which are necessary to control the ripening cycle of the cheese as far as possible. For every type of cheese, a specific combination of temperature and relative humidity must be maintained in the different storage rooms during the various stages of ripening.
Different types of cheese require different temperatures and relative humidity (RH) in the storage rooms. The climatic conditions are of great importance to the rate of ripening, loss of weight, rind formation and development of the surface flora (in Tilsiter, Romadur and others) – in other words to the total nature or characteristic of the cheese.
Cheeses with rinds, most commonly hard and semi-hard types, can be provided with a plastic emulsion or paraffin or wax coating. Rindless cheese is covered with plastic film or a shrinkable plastic bag.
- Cheeses of the Cheddar family are often ripened at low temperatures, 4–8°C, and a RH lower than 80%, as they are normally wrapped in a plastic film or bag and packed in cartons or wooden cases before being transported to the store. The ripening time may vary from a few months up to 8 – 10 months to satisfy the preferences of various consumers.
- Cheeses like Emmenthal may need to be stored in a “green” cheese room at 8 – 12 °C for some 3 – 4 weeks followed by storage in a “fermenting” room at 22 – 25 °C for some 6 – 7 weeks. After that the cheese is stored for several months in a ripening store at 8 – 12 °C. The relative humidity in all rooms is normally 85 – 90 %.
- Smear-treated types of cheese – Tilsiter, Havarti and others – are typically stored in a fermenting room for some 2 weeks at 14 – 16 °C and a RH of about 90%, during which time the surface is smeared with a special cultured smear mixed with a salt solution. Once the desired layer of smear has developed, the cheese is normally transferred to the ripening room at a temperature of 10 – 12 °C and a RH of 90 % for a further 2 – 3 weeks.
- Cheeses like Gouda and similar, may first be stored for a couple of weeks in a “green” cheese room at 10 – 12°C and a RH of some 75 %. After that a ripening period of about 3 – 4 weeks may follow at 12 – 18°C and 75 – 80% RH. Finally the cheese is transferred to a storage room at about 10 – 12°C and a relative humidity of about 75%, where the final characteristics are developed.
The values given for temperatures and relative humidity, RH, are approximate and vary for different sorts of cheese within the same group.